Super power
When hydrogen is burned in a fuel cell to generate electricity, the only waste product is water vapour
Orkney is suffering from an excess of electrons. Perched in the far north of Scotland, strong winds, fierce tides and waves generate more power than islanders can ever use – and it cannot easily be stored at scale. Nine thousand miles away in Singapore, Dr Adrian Fisher, Reader in Electrochemistry in the Department of Chemical Engineering and Biotechnology, says it is the perfect example of why electrochemistry’s time has come. “We need to find ways of using our surplus electrons,” he says.
Electrochemistry – using electricity to perform chemical reactions and create chemicals on demand – has been around since the 19th century, but the success of renewable energy has created a unique opportunity, because clean energy is erratic. Sometimes the wind blows, sometimes the sun shines – but if we don’t need that energy when it is produced, it can’t easily be stored. On Orkney, the solution is to use those extra electrons to liberate hydrogen from water (the electricity ‘splits’ the hydrogen from the oxygen). Pure hydrogen can then be stored and used in fuel cells.
This also has implications in space. It was just such a fuel cell that provided in-flight power to the 1969 Apollo 11 mission and, as Dr Fisher points out, the presence of ice on the far side of the moon could provide future missions with power, using electrochemistry. “Nasa is planning to go to the dark side of the moon by 2024. Where there is ice there is fuel – you can make hydrogen. And it means you are taking a localised resource to create something usable.”
When hydrogen is burned in a fuel cell to generate electricity, the only waste product is water vapour
But if hydrogen is clean, cheap and available, why don’t we use more of it? When hydrogen is burned or used in a fuel cell to generate electricity, the only byproduct is water vapour – there are no particulates or pollution. But storing hydrogen is expensive and complex. As the costs of renewable energy fall, it becomes economically more viable to produce hydrogen – thereby making a hydrogen economy more possible.
A hydrogen economy
In fact, hydrogen car prototypes have been around for decades – at 2019’s Frankfurt motorshow, BMW unveiled its latest model. However, switching is fraught with logistical headaches. “Infrastructure for hydrogen cells is not compatible with what is currently being rolled out for electric cars, but you can see prototypes on the market,” says Dr Minyu Zeng, a postdoctoral researcher who works alongside Dr Fisher. Germany now has a hydrogen train, San Francisco is welcoming a hydrogen ferry and there are thousands of hydrogen cars on California roads. “The supply chain of hydrogen is a huge project,” says Dr Zeng. “But it is time – the world is changing in how it produces energy.”
In fact, hydrogen is just one example of what you can do with electrochemistry. For the past six years, Dr Fisher and his team of chemists and engineers have been searching for cleaner ways to make chemicals, helping the industry become more sustainable. They have been collaborating in Cambridge and at CARES* (Cambridge Centre for Advanced Research and Education in Singapore Ltd, the University’s research centre in Singapore) on the Campus for Research Excellence and Technological Enterprise (CREATE). The Campus is funded by the National Research Foundation (NRF) of the Prime Minister’s Office in Singapore.
More than 100,000 chemical products are currently sold worldwide in a market worth around $3,500bn. Creating these products requires heat or the addition of another chemical to get going, whereas electrosynthesis works at low temperatures, and needs only a direct transfer of electrons, supplied by positive and negative electrodes dipped into the reaction mixture. No heat and no reagents mean lower costs, cleaner processes and greater precision. “Some chemicals are dangerous – it would be much better if we could make them locally,” says Dr Fisher. The ultimate vision is for chemicals produced on demand, on site, resulting in less waste and lower transport costs.
There are challenges, however. “Although most aluminium today is already made electrochemically, I wouldn’t want to say you could make everything this way,” says Dr Fisher. Logistics aren’t always straightforward. “Our catalyst is connected to an electrical supply, and that’s not quite as easy as just putting a chemical catalyst into a large reactor and allowing it to do its stuff,” he says. “In principle you can do a huge amount of chemistry electrochemically, but, historically, the cost of electrons has limited progress. And the technology is not yet as advanced as it needs to be to replace current large-scale manufacture.”
But ultimately, he says, all chemistry is really electrochemistry – moving electrons and making and breaking bonds. “This could be a completely different way of looking at the production of chemicals,” says Dr Fisher. “That’s our role in the Singapore centre. If you can take local feedstocks, and make chemicals locally and when you need them, you don’t have to ship them around the world or store them.”
The power of data
And Dr Zeng is working on other potential applications. “Water is two hydrogen atoms plus one oxygen. But what if you have two molecules, each H2O2? That’s hydrogen peroxide – an important chemical that is used as a disinfectant and as a reagent in many chemical processes. If you can generate electricity and produce a useful product, it’s a win-win.” However, there have been problems with this process, including weak concentration of H2O2, corroded electrodes and low levels of electricity. “But it still has potential in particular applications such as in the paper-making industry – or in cleaning products,” says Dr Zeng.
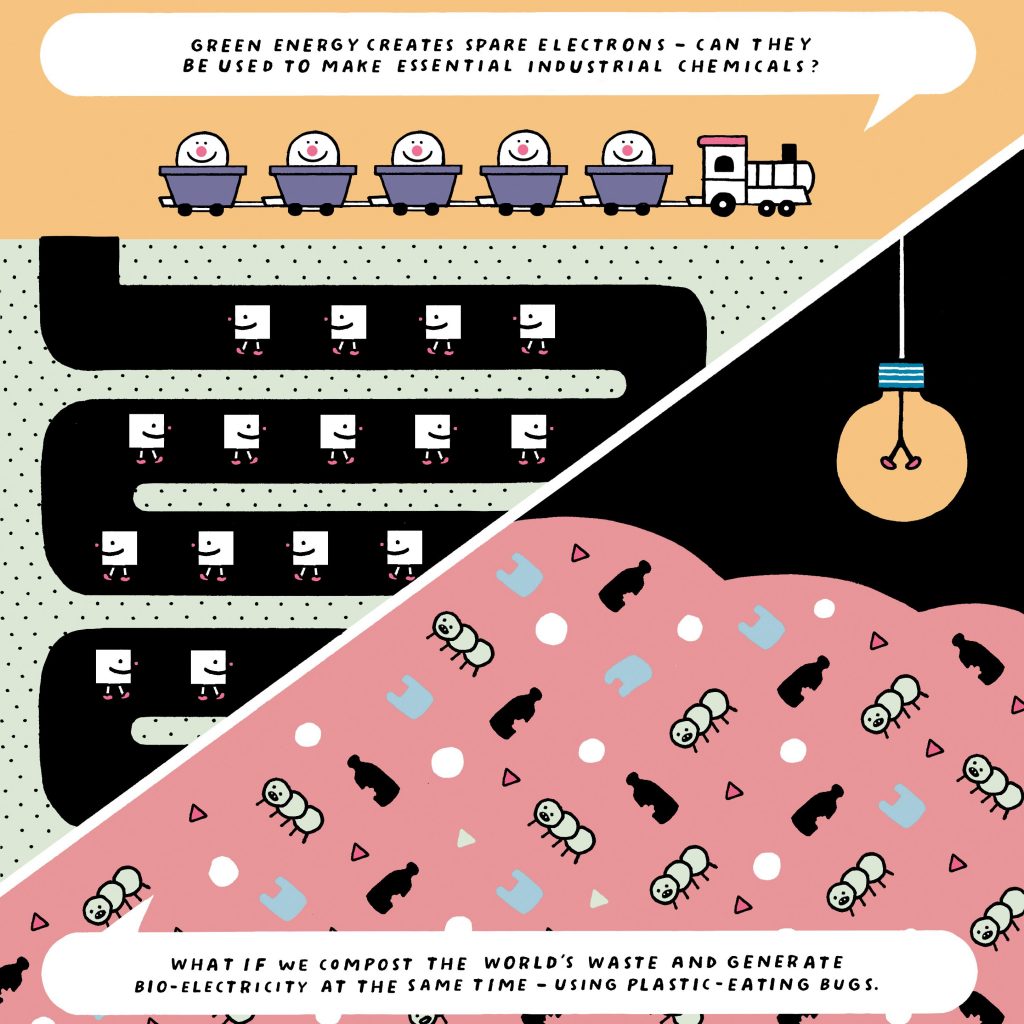
He is also working on technology that harnesses the power of data – such as looking for patterns in the supply of solar and wind energy, and demand for chemicals. “If you use artificial intelligence to process this data, you might find a pattern. You could feed this information to a chemical-generation platform, so we would know what chemicals to produce, when and in what amounts.” He aims to pilot this idea within the coming months. “Our goal is to make a platform that could help generate multiple chemicals.”
Plastic composters
When algae photosynthesise, they create a flow of electrons, some of which are transported outside the cell. The aim is to capture these electrons on
a biophotovoltaic platform
Back in Cambridge, Dr Fisher’s team is investigating whether electrochemistry can clean up waste supplies and create products that could be used to create energy. In 2016, Japanese scientists discovered tiny bugs found in landfill sites that could literally eat plastic, and chemical engineer, Dr Aazraa Oumayyah Pankan, is currently trying to recreate this process in the lab to see how it works. However, breaking down the plastic is not actually her ultimate aim. When microalgae photosynthesise, they create a flow of electrons, some of which are transported outside the cell. And so, as Dr Pankan explains, her true purpose is to “capture these [electrons] on a biophotovoltaic platform – a biological solar cell – to generate electricity”. Once the team has identified the single molecules that are products of the broken-down plastic, Dr Pankan wants to harvest energy from them, by feeding them to low-cost and widely available microalgae (cyanobacteria). “We want to see if we can use these biodegradable products [monomers] to generate bioelectricity.”
And if you could harness these plastic composters to provide electricity in rural communities, it would solve two problems in one. It’s fundamental science: improving these cells’ ability to export more electrons rather than use them for photosynthesis. “We’re trying to make the whole process more effective and efficient,” she says.
Bioelectricity is also being generated from tropical microalgae in Malaysia. Working with scientists from the University of Malaya, the team has been putting microbes to work, using them to clean up waste water from the palm-oil industry – and providing power. “We showed that these microalgae fed on diluted palm-oil mill effluent, photosynthesised, and generated bioelectricity at the same time,” says Dr Pankan.
This, she explains, is potentially a cheap and sustainable system, and is currently being tested in a pilot project in Malaysia. “You just need microorganisms, which are basically the biocatalyst. Add some nutrients, some water and light energy. The bacteria use carbon dioxide to photosynthesise, and the only waste product of the whole system is water.”
New ways of working
Devising new ways of working is critical if nations are to meet pledges to cut carbon emissions and address the urgency of the crisis, says Dr Fisher. “There are sporadic examples emerging of new processes that do work and make sense – on Orkney, for instance.” Collaborating with scientists around the work, he likens his team to pieces of a jigsaw puzzle. “You need a broad range of groups with different expertise to be able to make progress. We are competing against processes that have existed for 100 years or more.”
He firmly believes innovation can lead to new technological solutions for manufacturing chemicals. “None of us really knows how to bring this new technology to market, but we do know that the industry won’t be operating in the same way in 30 years’ time.” As Dr Fisher sees it, opportunities offered by electrochemistry are vast. “This could be a completely different way of approaching the chemicals industry – and that’s urgently needed if we are to tackle CO2 emissions and meet our climate targets.”
*This research is supported by the National Research Foundation (NRF), Prime Minister’s Office, Singapore under its Campus for Research Excellence and Technological Enterprise (CREATE) programme.